 To prevent this from happening, cells control how many ribosomal proteins they make, so there are just enough to form the ribosomes the cell needs at any given time. The ribosomes themselves are made up of several proteins, some of which are also prone to clumping as they are being produced. Some newly-made proteins are prone to forming clumps, so cells have other proteins known as chaperones that ensure these clumps do not form. The newly-made proteins then have to travel to a specific location in the cell to perform their tasks. Machines called ribosomes inside the cells use template molecules called messenger RNAs (or mRNAs for short) to produce proteins. Living cells are packed full of molecules known as proteins, which perform many vital tasks the cells need to survive and grow. 
Taken together, we have uncovered an unprecedented regulatory mechanism that adjusts the de novo synthesis of Rpl3 and Rpl4 to their actual consumption during ribosome assembly and, thereby, protects cells from the potentially detrimental effects of their surplus production. Notably, deregulated expression of Rpl3 and Rpl4 leads to their massive aggregation and a perturbation of overall proteostasis in cells lacking the E3 ubiquitin ligase Tom1. In both cases, negative regulation of mRNA levels occurs when the availability of the dedicated chaperone is limited and the nascent ribosomal protein is instead accessible to a regulatory machinery consisting of the nascent-polypeptide-associated complex and the Caf130-associated Ccr4-Not complex. Here, we show that the co-translational recognition of Rpl3 and Rpl4 by their respective dedicated chaperone, Rrb1 or Acl4, reduces the degradation of the encoding RP元 and RPL4 mRNAs in the yeast Saccharomyces cerevisiae. Recent evidence has highlighted that dedicated chaperones protect individual, unassembled ribosomal proteins on their path to the pre-ribosomal assembly site. Supplying equimolar amounts of assembly-competent ribosomal proteins is complicated by their aggregation propensity and the spatial separation of their location of synthesis and pre-ribosome incorporation. Our pages contain various quotes with which our editorial team does not always agree.The biogenesis of eukaryotic ribosomes involves the ordered assembly of around 80 ribosomal proteins. See also: Alpha male, Origin of life, Deamination, Evolutionary Clock, Neurula Her father, who'd gained considerable wealth trading with Japan and China, stayed behind. 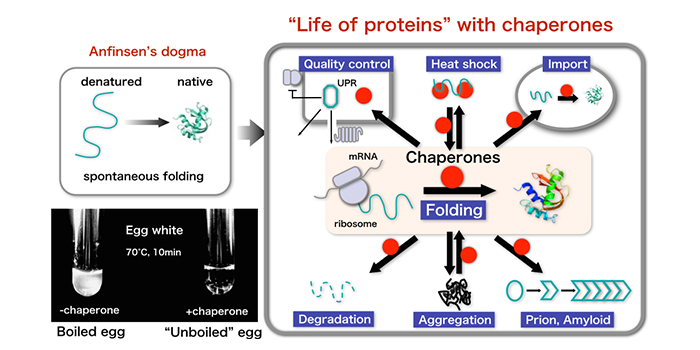
The talented young woman sailed from her home in Portland, Oregon, to Florence, Italy, d by her mother. ERp29, protein disulfide isomerase, BiP/Grp78, calnexin, etc.).īut Capecchi's grandmother, Lucy Dodd, was determined to become a painter. The nascent protein is folded in the ER by the proteins (e.g. 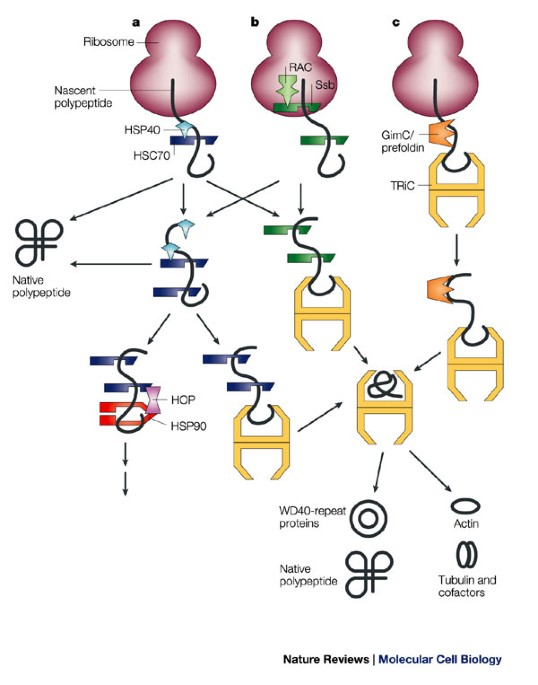
It is the job of a class of proteins called s to bind to the growing protein chains as they are synthesized by ribosomes and prevent aggregates from forming or the proteins from folding incorrectly before they have been fully synthesized.Īn example of an important endoplasmic reticulum resident protein is the protein known as BiP (formally: the immunoglobulin-binding protein), which identifies other proteins that have been improperly built or processed and keeps them from being sent to their final destinations. In the absence of steroid hormone, the receptors cling together in a complex called aporeceptor complex, which also contains proteins (also known as heatshock proteins or Hsps). CNX/CRT assist in protein folding in consort with glycosylation. Prominent molecular s of the rough ER include binding immunoglobulin protein (BiP), Calnexin (CNX) and Calreticulin (CRT). The ER signal sequences are recognized by proteins which guide the synthesizing ribosomes to the rough ER where translocation of the protein sequence occurs in a protein complex named the translocon. S are protective molecules of body's proteins, they have helped to folding process in proteins and prevented of their aggregation, therefore they have more useful function in prevention of many diseases like Alzheimer and diabetes type II. : Any cellular protein that binds to an unfolded or partially folded target protein to prevent misfolding, aggregation, and/or degradation of it. If you know of any terms that have been omitted from this glossary that you feel would be useful to include, please send details to the Editorial Office at GenScript.Ī protein that facilitates the folding of other proteins or assembly of multi-protein complexes.Ĭollective term for two types of proteins that prevent misfolding of a target protein (molecular s) or actively facilitate its proper folding (chaperonins). Please help improve it or discuss these issues on the talk page. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
